Signaling Properties of Structurally Diverse Kappa Opioid Receptor Ligands: Toward in Vitro Models of in Vivo Responses | ACS Chemical Neuroscience
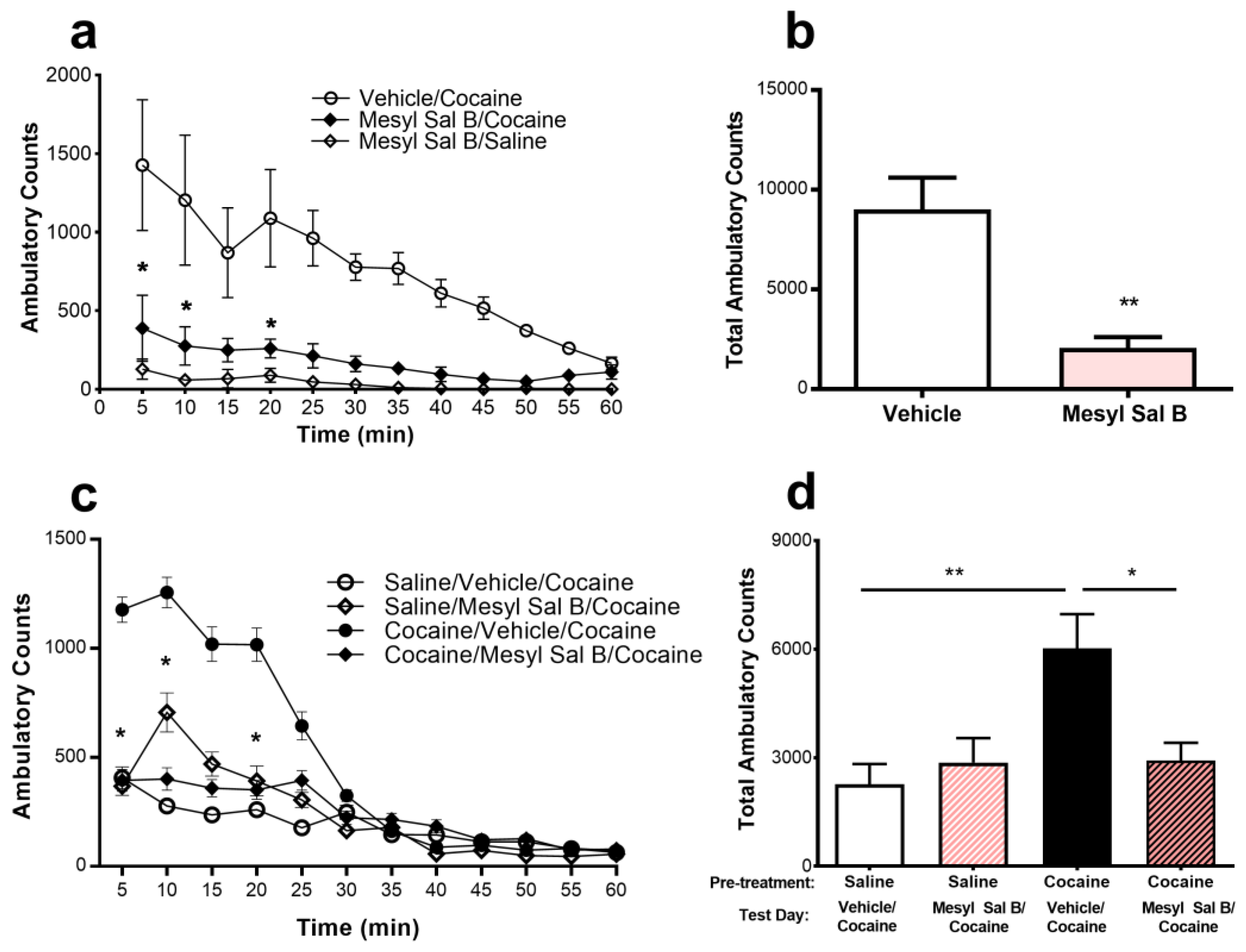
Molecules | Free Full-Text | Kappa Opioid Receptor Agonist Mesyl Sal B Attenuates Behavioral Sensitization to Cocaine with Fewer Aversive Side-Effects than Salvinorin A in Rodents | HTML
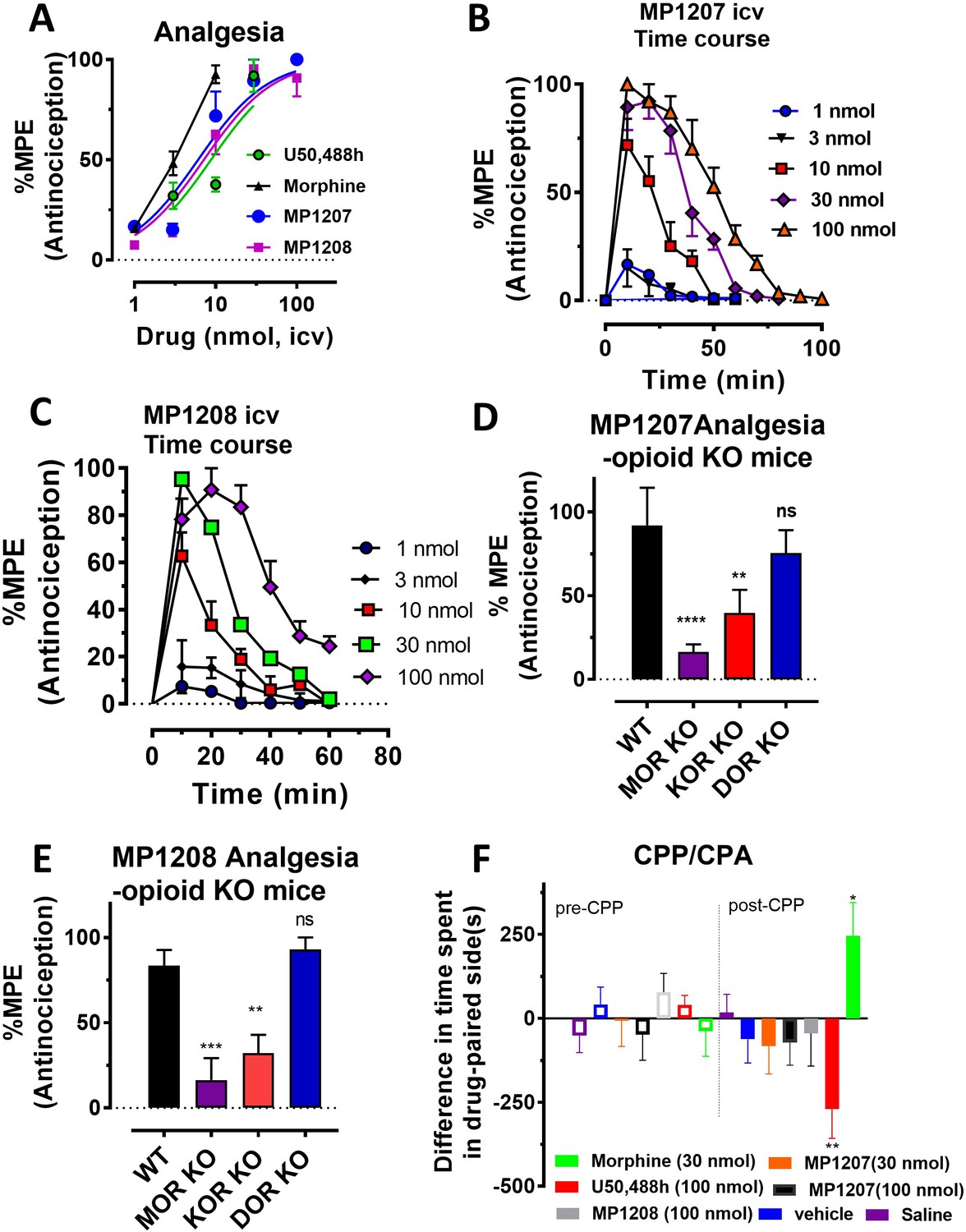
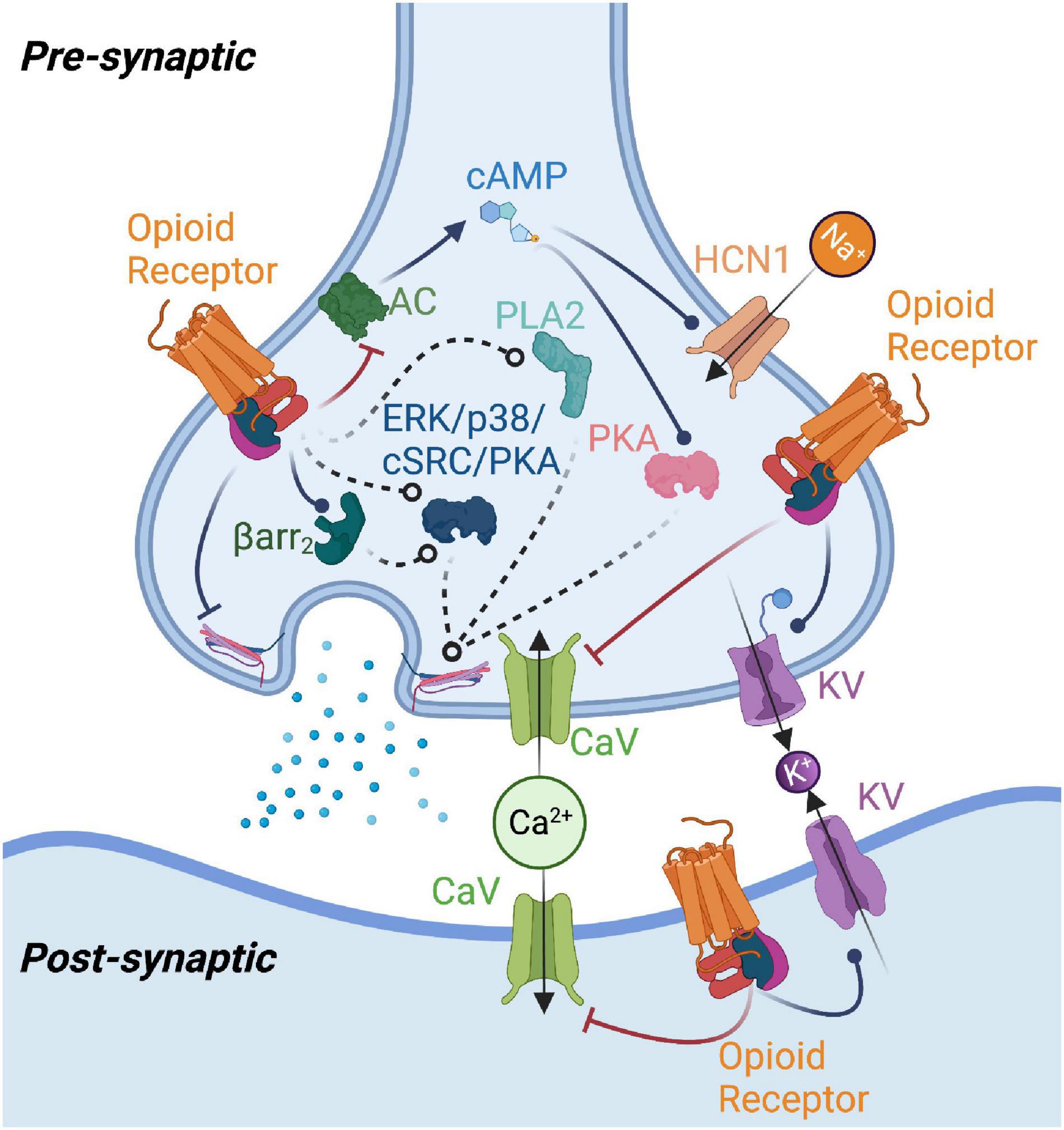
Controlling opioid receptor functional selectivity by targeting distinct subpockets of the orthosteric site | eLife
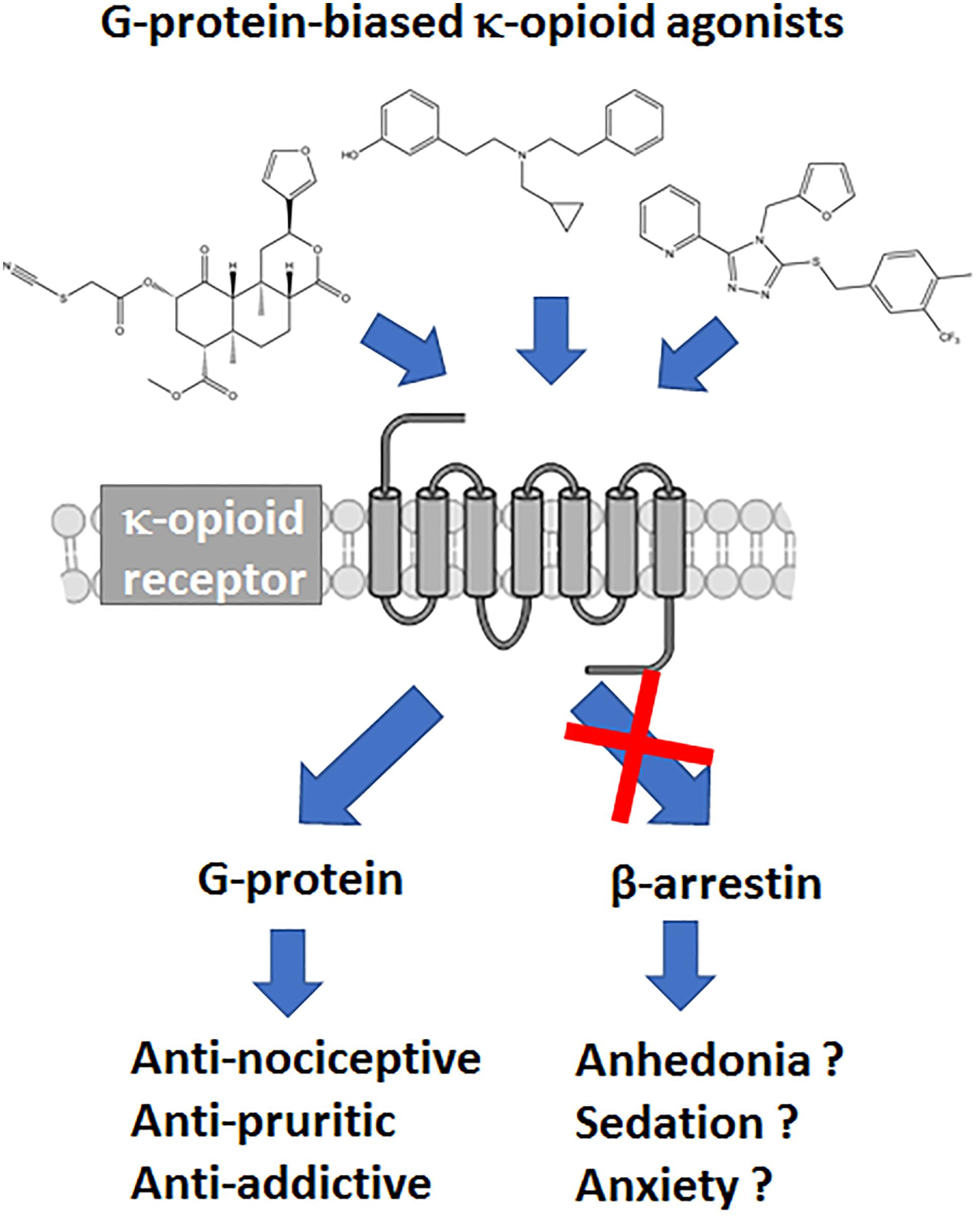
Frontiers | A Review of the Therapeutic Potential of Recently Developed G Protein-Biased Kappa Agonists

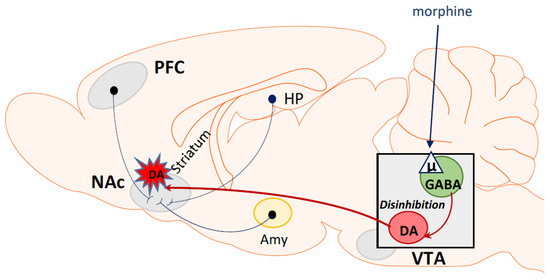
κ Opioid Receptor Antagonism and Prodynorphin Gene Disruption Block Stress-Induced Behavioral Responses | Journal of Neuroscience
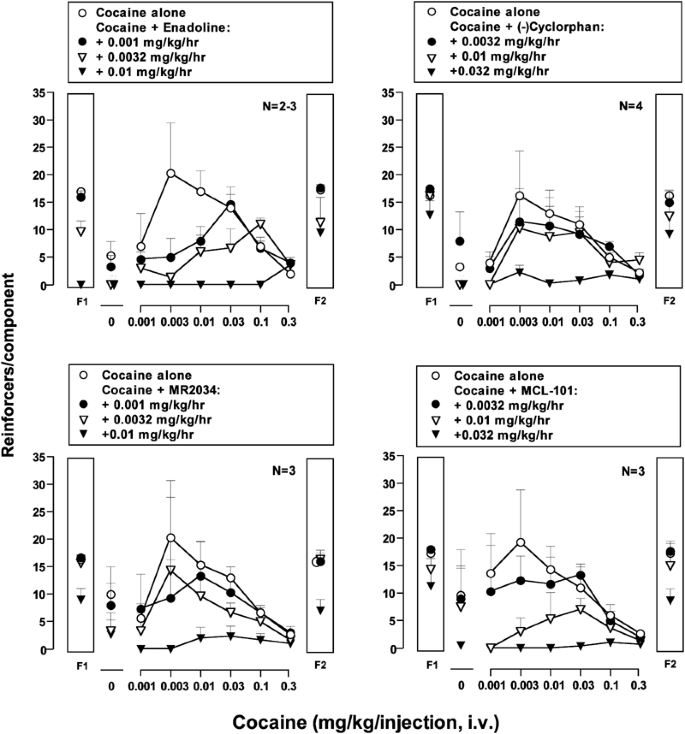
Effects of Mixed-Action κ/μ Opioids on Cocaine Self-Administration and Cocaine Discrimination by Rhesus Monkeys | Neuropsychopharmacology

Critical Assessment of G Protein-Biased Agonism at the μ-Opioid Receptor: Trends in Pharmacological Sciences

Effects of Kappa Opioid Receptor Agonists on Fentanyl vs. Food Choice in Male and Female Rats: Contingent vs. Non-Contingent Administration | bioRxiv

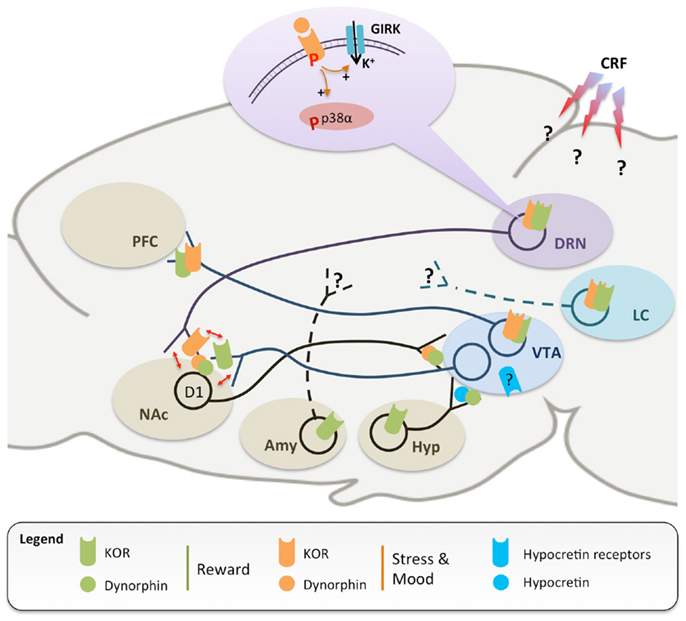
Kappa Opioid Receptors Regulate Stress-Induced Cocaine Seeking and Synaptic Plasticity - ScienceDirect

The Role of Dynorphin and the Kappa Opioid Receptor in the Symptomatology of Schizophrenia: A Review of the Evidence - Biological Psychiatry

Deep brain stimulation of the nucleus accumbens shell attenuates cocaine withdrawal but increases cocaine self-administration, cocaine-induced locomotor activity, and GluR1/GluA1 in the central nucleus of the amygdala in male cocaine-dependent rats -

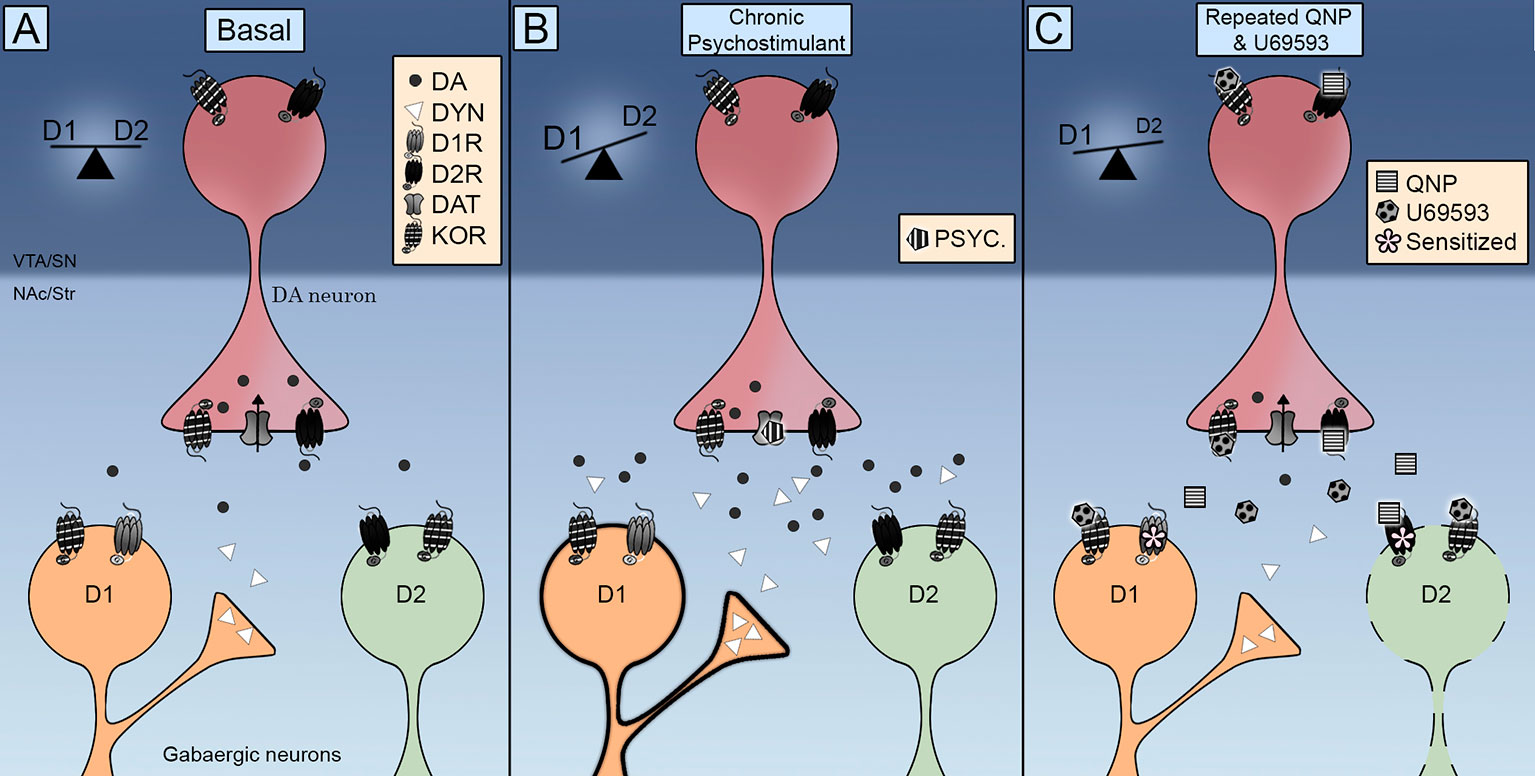
κ Opioid Receptors in the Nucleus Accumbens Shell Mediate Escalation of Methamphetamine Intake | Journal of Neuroscience

Effects of the Kappa-opioid Receptor Agonist, U69593, on the Development of Sensitization and on the Maintenance of Cocaine Self-administration | Neuropsychopharmacology
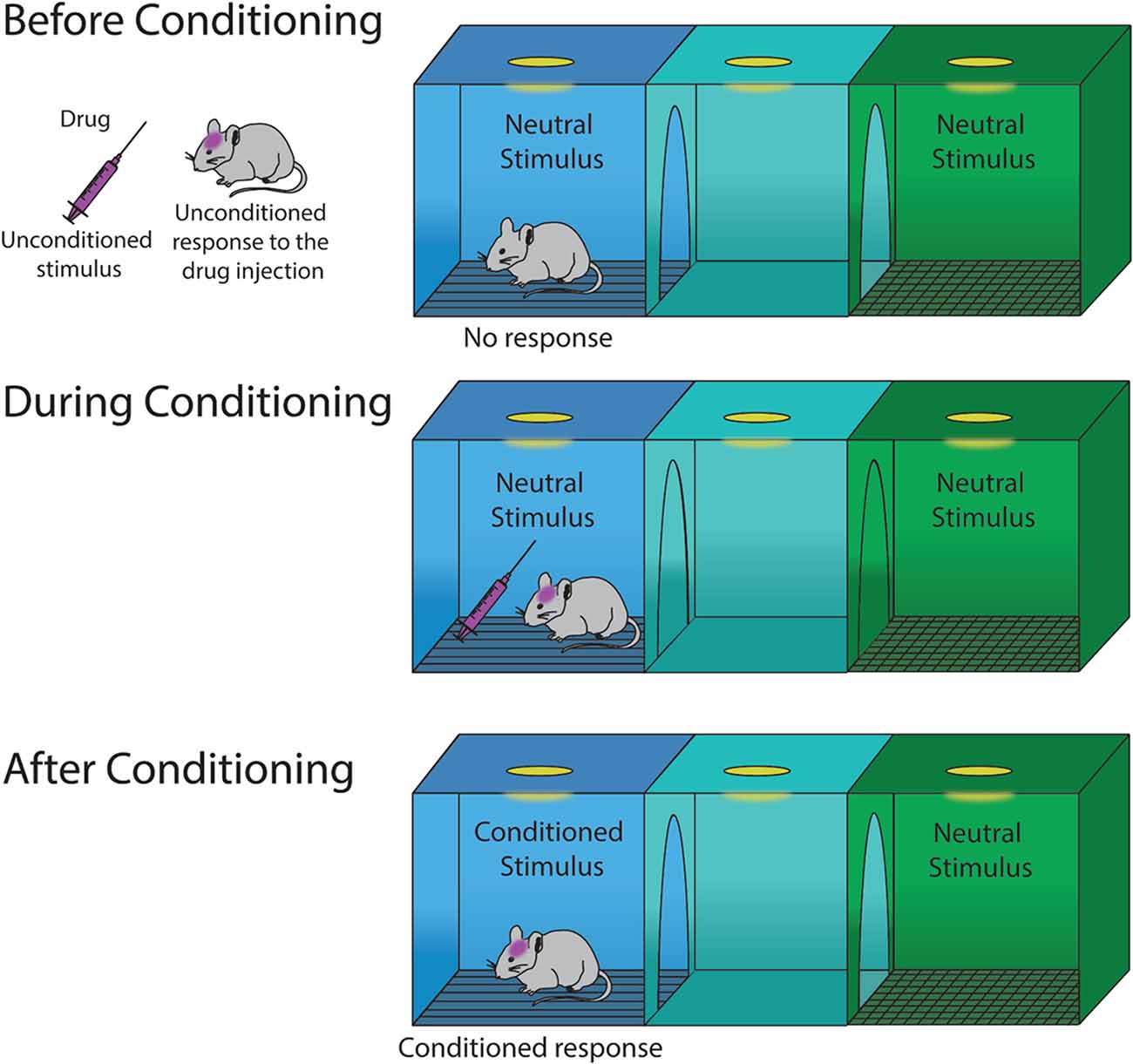
Frontiers | Drug-Induced Conditioned Place Preference and Its Practical Use in Substance Use Disorder Research
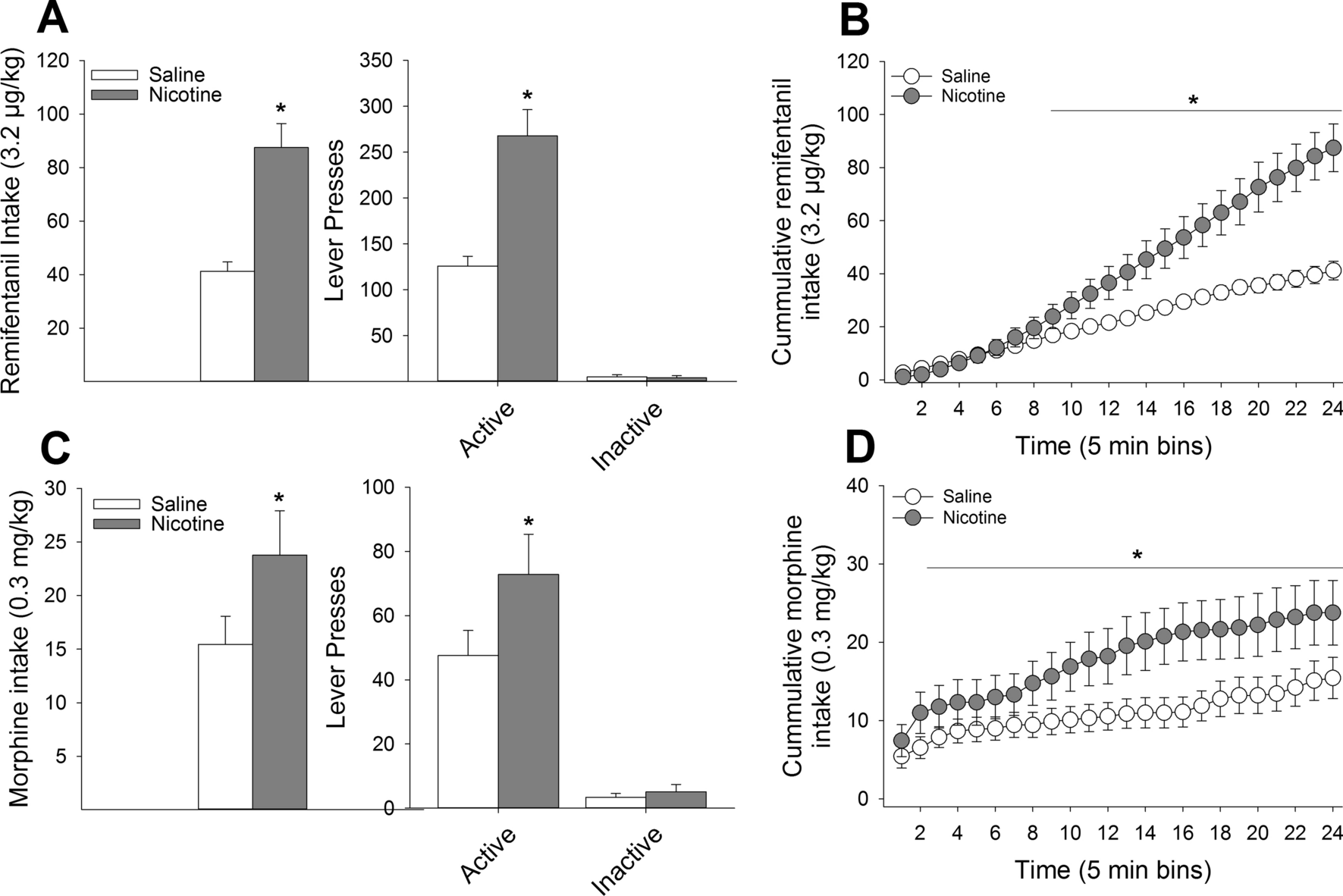
Systemic nicotine enhances opioid self-administration and modulates the formation of opioid-associated memories partly through actions within the insular cortex | Scientific Reports

Effects of Kappa Opioid Receptor Agonists on Fentanyl vs. Food Choice in Male and Female Rats: Contingent vs. Non-Contingent Administration | bioRxiv
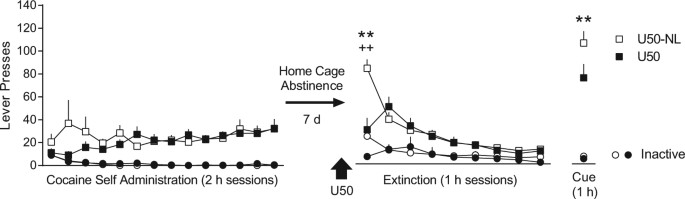
A single, extinction-based treatment with a kappa opioid receptor agonist elicits a long-term reduction in cocaine relapse | Neuropsychopharmacology

Interactions between Kappa Opioid Agonists and Cocaine: Preclinical Studies - MELLO - 2000 - Annals of the New York Academy of Sciences - Wiley Online Library

A single, extinction-based treatment with a kappa opioid receptor agonist elicits a long-term reduction in cocaine relapse | Neuropsychopharmacology

Self-Administered Heroin and Cocaine Combinations in the Rat: Additive Reinforcing Effects—Supra-Additive Effects on Nucleus Accumbens Extracellular Dopamine | Neuropsychopharmacology
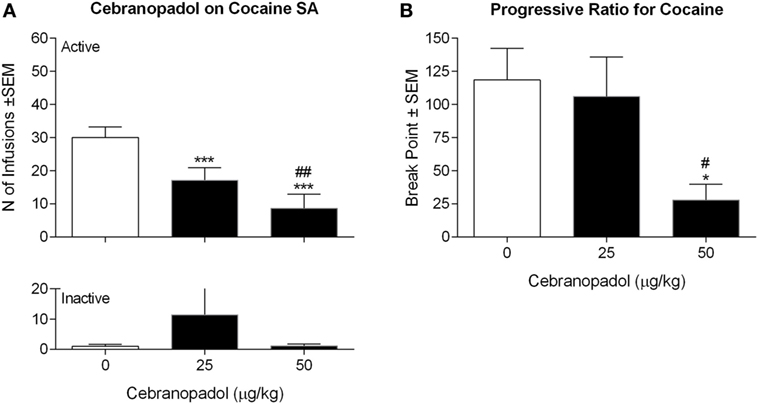
Frontiers | Cebranopadol, a Mixed Opioid Agonist, Reduces Cocaine Self- administration through Nociceptin Opioid and Mu Opioid Receptors