
Regioselective Synthesis of Pyrroles from Alkyne‐Isocyanide Click Reactions: An Angle Strain‐Induced Bond Migration Approach - George - 2016 - Advanced Synthesis & Catalysis - Wiley Online Library

Scheme 2. Synthesis of pyrroles from nitroarenes in the presence of... | Download Scientific Diagram
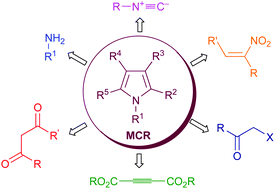
Recent advances in the synthesis of pyrroles by multicomponent reactions - Chemical Society Reviews (RSC Publishing)

Convenient synthesis of polysubstituted pyrroles and symmetrical and unsymmetrical bis-pyrroles catalyzed by H3PW12O40 - ScienceDirect
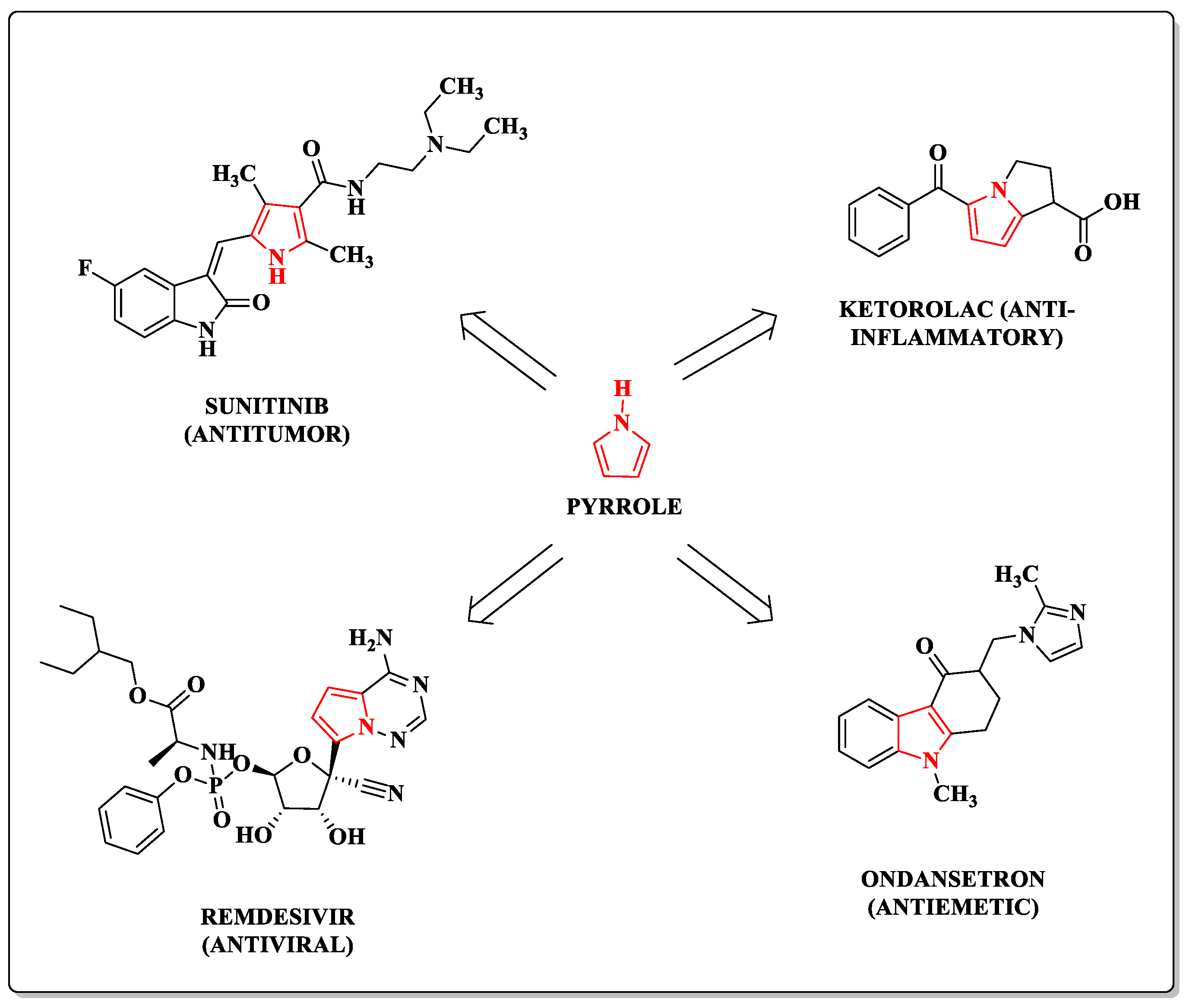
Pharmaceuticals | Free Full-Text | Pyrroles as Privileged Scaffolds in the Search for New Potential HIV Inhibitors
![Synthesis of Pentasubstituted 2-Aryl Pyrroles from Boryl and Stannyl Alkynes via One-Pot Sequential Ti-Catalyzed [2+2+1] Pyrrole Synthesis/Cross Coupling Reactions | Catalysis | ChemRxiv | Cambridge Open Engage Synthesis of Pentasubstituted 2-Aryl Pyrroles from Boryl and Stannyl Alkynes via One-Pot Sequential Ti-Catalyzed [2+2+1] Pyrrole Synthesis/Cross Coupling Reactions | Catalysis | ChemRxiv | Cambridge Open Engage](https://chemrxiv.org/engage/api-gateway/chemrxiv/assets/orp/resource/item/60c74b22f96a004032287583/largeThumb/synthesis-of-pentasubstituted-2-aryl-pyrroles-from-boryl-and-stannyl-alkynes-via-one-pot-sequential-ti-catalyzed-2-2-1-pyrrole-synthesis-cross-coupling-reactions.jpg)
Synthesis of Pentasubstituted 2-Aryl Pyrroles from Boryl and Stannyl Alkynes via One-Pot Sequential Ti-Catalyzed [2+2+1] Pyrrole Synthesis/Cross Coupling Reactions | Catalysis | ChemRxiv | Cambridge Open Engage
![Molecules | Free Full-Text | Synthesis of Multi-Substituted Pyrrole Derivatives Through [3+2] Cycloaddition with Tosylmethyl Isocyanides (TosMICs) and Electron-Deficient Compounds | HTML Molecules | Free Full-Text | Synthesis of Multi-Substituted Pyrrole Derivatives Through [3+2] Cycloaddition with Tosylmethyl Isocyanides (TosMICs) and Electron-Deficient Compounds | HTML](https://www.mdpi.com/molecules/molecules-23-02666/article_deploy/html/images/molecules-23-02666-sch001-550.jpg)
Molecules | Free Full-Text | Synthesis of Multi-Substituted Pyrrole Derivatives Through [3+2] Cycloaddition with Tosylmethyl Isocyanides (TosMICs) and Electron-Deficient Compounds | HTML
![Synthesis of pyrroles via ruthenium-catalyzed nitrogen-transfer [2 + 2 + 1] cycloaddition of α,ω-diynes using sulfoximines as nitrene surrogates | Communications Chemistry Synthesis of pyrroles via ruthenium-catalyzed nitrogen-transfer [2 + 2 + 1] cycloaddition of α,ω-diynes using sulfoximines as nitrene surrogates | Communications Chemistry](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fs42004-018-0022-2/MediaObjects/42004_2018_22_Fig1_HTML.jpg)
Synthesis of pyrroles via ruthenium-catalyzed nitrogen-transfer [2 + 2 + 1] cycloaddition of α,ω-diynes using sulfoximines as nitrene surrogates | Communications Chemistry
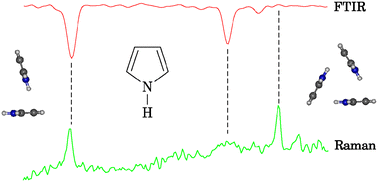
N–H⋯π interactions in pyrroles: systematic trends from the vibrational spectroscopy of clusters - Physical Chemistry Chemical Physics (RSC Publishing)

Synthesis of 2,3,4-trisubstituted pyrroles via a facile reaction of vinyl azides and tosylmethyl isocyanide

The Oxidation of Pyrrole - Howard - 2016 - Chemistry – An Asian Journal - Wiley Online Library

Substrate Controlled Regioselective Bromination of Acylated Pyrroles Using Tetrabutylammonium Tribromide (TBABr3) | The Journal of Organic Chemistry

Organocatalytic Enantioselective Friedel‐Crafts Alkylation Reactions of Pyrroles - Gaviña - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library

Life | Free Full-Text | The Abiotic Formation of Pyrrole under Volcanic, Hydrothermal Conditions—An Initial Step towards Life's First Breath?

Pyrrole, 有機体である, compound. Pyrroles, chlorins, heme, porphyrins, macrocycles, heterocyclic, 芳香がする, bacteriochlorins, pyrrole | CanStock





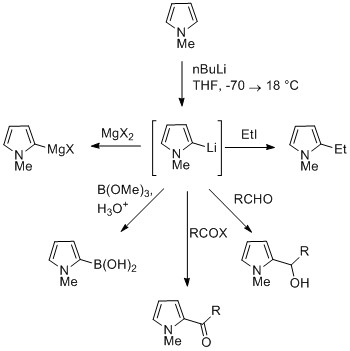




![Pyrroles [Non-Halogenated Heterocyclic Building Blocks] | TCI EUROPE N.V. Pyrroles [Non-Halogenated Heterocyclic Building Blocks] | TCI EUROPE N.V.](https://www.tcichemicals.com/medias/H0826.jpg?context=bWFzdGVyfHJvb3R8MzQwNzZ8aW1hZ2UvanBlZ3xoMzYvaGE3Lzg5MzA4OTc5NTI3OTgvSDA4MjYuanBnfGMyMGE3MWVjZDM0NWRhOTAzNTA3MTQ0MjljMDM3MTkzYWQwZDg1NWEyZDQyMDEzMjY1MjZjNTI5Y2MwOTNjZjc)
